 Hint: Substitute the given function into the relation c v = T(∂ s/∂ T) v. Use the results of Example 11.1 to show that a material that has an entropy function of the form s( T, v) = A + B(ln T) + C(ln v), where A, B, and C are constants, has a constant volume specific heat given by c v = B. 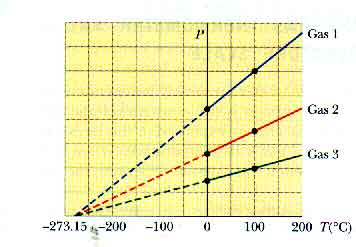
The following exercises are designed to strengthen your understanding of the thermodynamics and the mathematics of the material presented in this part of the chapter. (2.35) S nĮquilibrium reaction rate for species i WĬonserved scalar or Shvab-Zeldovich function γ Kinetic rate factor for species i-see eqn. Stoichiometric mass ratio of oxidant to fuel r Radius to where ζbar is half its value on the axis L Rįavre probability density function, eqn. 
From Bilger and Beck 36 with permission of The Combustion Institute. Normalized peak nitric oxide mass fraction and emission index, E = lim ξ → 0 ( Y N O / ξ ), for turbulent diffusion flames of hydrogen jets into still air under similarity conditions. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
